New study finds keys to brain cancer growth, may help improve treatments
The findings could help doctors predict and slow brain tumor regrowth, leading to more personalized treatments for glioblastoma.
An interdisciplinary team of researchers led by Noemi Andor and Ana Gomes at Moffitt Cancer Center, and Parag Katira, a mechanical engineering professor at San Diego State University, has discovered how brain cancer cells grow and change based on their environment, which could help doctors predict how tumors respond to treatment.
Their NIH-supported study, published in Cancer Research, looked at glioblastoma, the most aggressive type of brain tumor. Their research focused on ploidy, which refers to the number of chromosomes in a cell. Researchers discovered cancer cells with different ploidy levels react differently to oxygen and nutrients in the brain, which affects how quickly the tumor returns after surgery.
“Our findings show that the resources available in the brain influence how glioblastoma cells evolve,” said Andor, assistant member in the Integrated Mathematical Oncology Department at Moffitt and co-lead author of the study. “This could help us predict tumor growth and personalize treatment plans.”
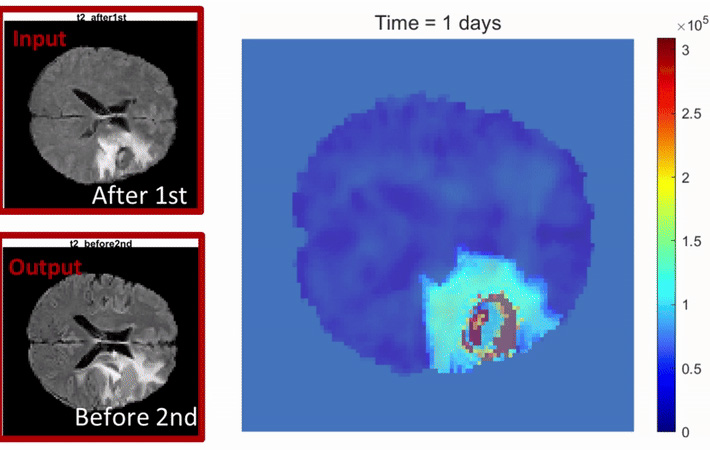
The study used a new mathematical model called the Stochastic State-Space Model of the Brain. This model combines patient data, medical images and lab research to predict how glioblastoma cells grow, move, and respond to treatments.
“The mathematical model is a low computational cost, high-dimensional model that uses the patient's clinical data to identify the hidden history of tumor evolution and how it was shaped by the tumor environment,” said Katira. “This has key implications in understanding the factors driving tumor progression and response to therapeutic interventions.”
The validation and integration of this mathematical model with clinical observations and experimental analysis was facilitated by a large collaborative effort from trainees and principal investigators across different fields of oncology, defining a pipeline for multimodal data integration and prediction of glioblastoma progression.
Using this pipeline, the team studied brain scans from glioblastoma patients to track tumor regrowth. They found that tumors with different ploidy levels grow back at different speeds, depending on where they are in the brain. “This research could help us find new ways to slow down tumor growth by targeting the energy supply that cancer cells rely on,” Andor said.
Key findings suggest some glioblastoma cells accelerate their switch to using sugar for energy more quickly than others, with this transition being influenced by the availability of oxygen. The levels of oxygen and nutrients in the brain play a significant role in determining how tumors grow and how they may recur after treatment. A deeper understanding of how tumors utilize energy could potentially help doctors make more informed treatment decisions.
“The implications of our work could be significant not only in glioblastoma. In a high-level analysis we observed an association between resource availability in different tissues and the ploidy of tumors emergent from those sites. This potentially indicates that energy supply and cancer genome evolution are tightly connected across cancers,” said Zuzanna Nowicka, a researcher from the Medical University of Lodz, Lodz, PL, and co-first author of the study.
This study was supported by the National Institutes of Health (1R37CA266727-01A1, 1R21CA269415-01A1 and R03CA259873-01A1 to N.A.) and by the Moffitt Cancer Center Evolutionary Therapy Center of Excellence (IMO workshop Pilot Award).



